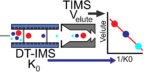
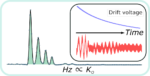
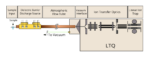
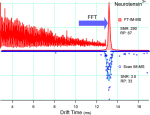
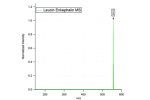
Just accepted in JASMS, we argue in this critical insight that in order to enable the development of new-generation CCS […]
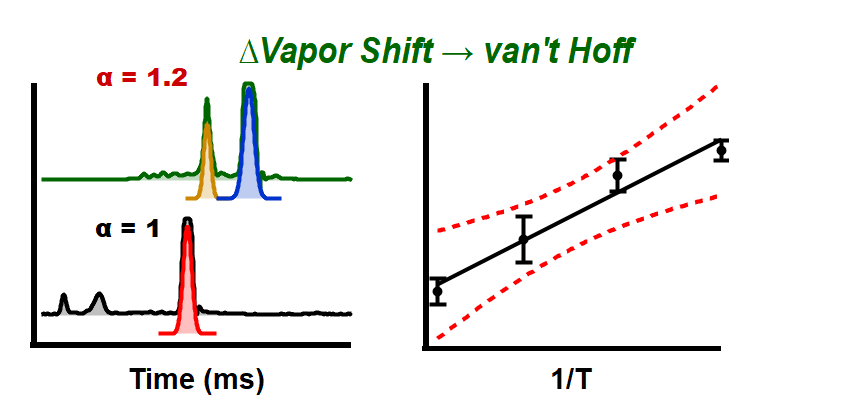
Just accepted in JASMS: Vapor assisted mobility shift measurements were made with atmospheric pressure drift-tube ion mobility–mass spectrometry (IM–MS) to […]
2 group members, Elvin and Cameron, have presentations at the virtual ISIMS on twitter this year. Go check out what […]
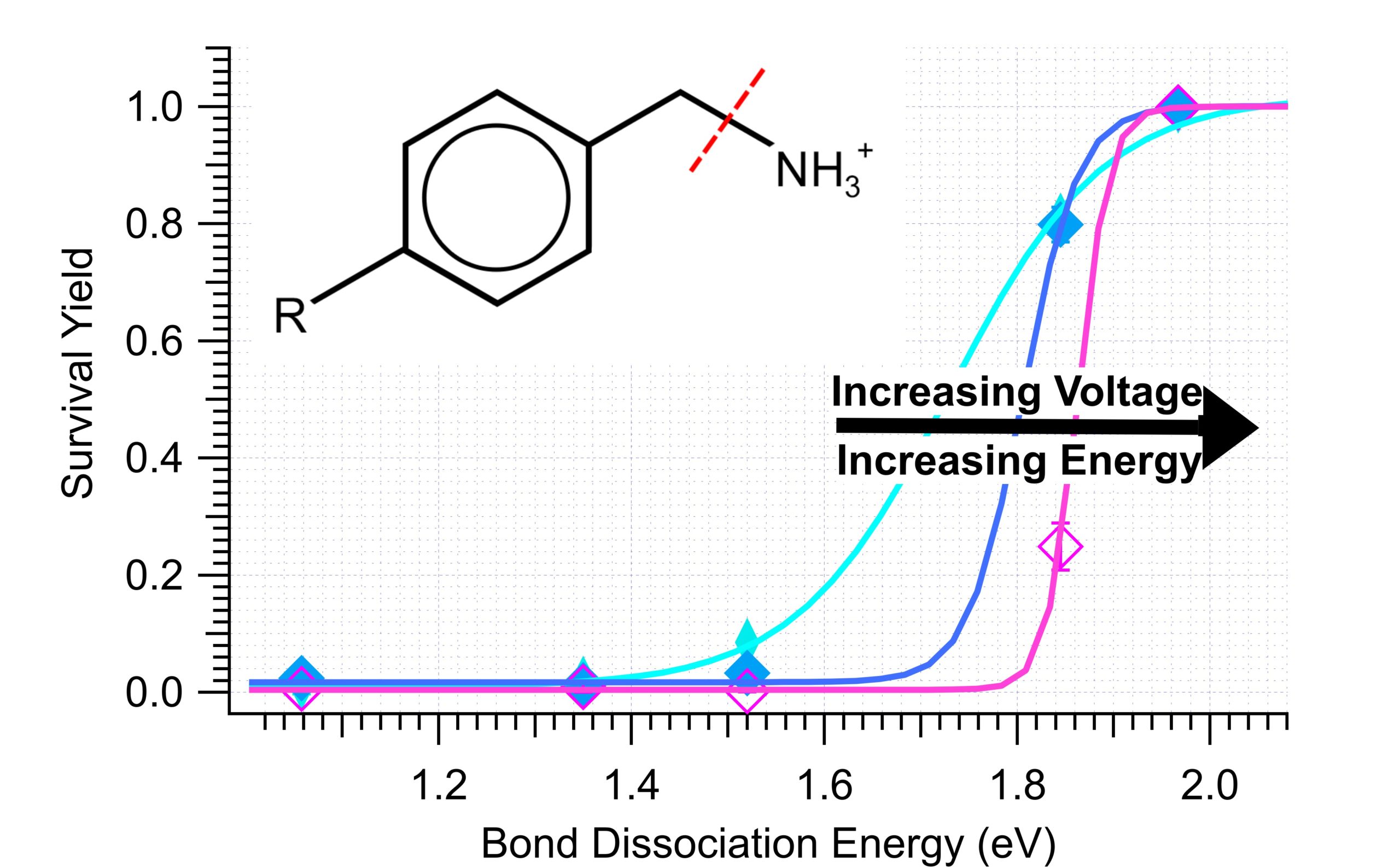
Just accepted in JASMS, we evaluate of TIMS source conditions to determine an effective energy for different voltages applied to […]
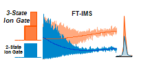
Just published in International Journal for Ion Mobility Spectrometry, we compare the ability of the tri-state ion shutter (3S-IS) to the […]
Pearl Kwantwi-Barima successfully defended her Ph.D. dissertation this semester. Congrats Pearl!
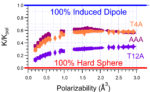
Our latest manuscript was just accepted in Analytical Chemistry. We use a set of standards to discuss the implications of […]
Just accepted in Analyst, we’ve improved on using our tri-shutter grid ion gates by implementing the tri-state pulsing to increase […]
While trapped ion mobility spectrometry (TIMS) provides excellent separation capability as an ion mobility technique, one major drawback is the […]
In a standard single averaged, drift tube ion mobility spectrometry (IMS) experiment, typically less than 1% of the ions are […]
A brief, condensed tutorial of IMS calculations and simulation is provided in the following jupyter notebook. This example code is […]
Previously, we reported an approach to quantify the energetics of association between neutral drift gas modifiers and two common chemical […]
Recently we’ve been working on the design and construction of a new instrument build centering around a quadrupole mass spectrometer […]
Following our initial experiments using ion trap mass spectrometry and atmospheric flow tube sampling (AFT-MS) for alkylphosphonic acid detection, we […]
For those that are interested, here is the spreadsheet used in the ASMS 2018 short course. Thank Dr. Bill Siems […]
Atmospheric flow tube-mass spectrometry (AFT-MS) first emerged in 2012 as an ambient vapor sampling technique developed by Ewing et al. […]
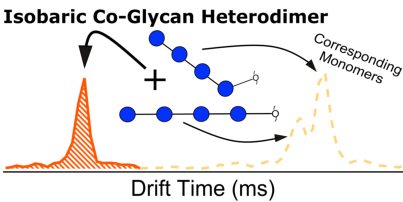
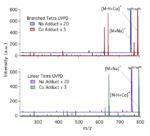
Continuing along our previous work to characterize certain cationized glycans with a variety of metals and with cobalt to enhance […]
Following up on previous post, we’ve finally released a major update to the Open Source IMS Initiative. Appearing now […]
Ion gating remains a critical aspect of drift tube IMS experiment and a range of clever approaches have been used […]
Ultraviolet photodissociation (UVPD) has had its application directed primarily at biomolecule analysis, but with a common drawback of low fragment […]
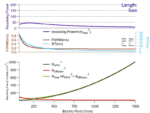
Austen has recently assembled an animation demonstrating the effect of increasing drift tube length on resolving power, calculated from peak […]
Using a combination of Fourier transform ion mobility and ion trap mass spectrometry Kelsey demonstrated the degree to which different metal […]
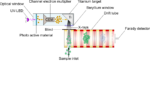
Outside of an ionization source and a Faraday plate, a drift tube IMS system is fundamentally comprised of 5 primary […]
We are pleased to report the publication of our work outlining the effective coupling of a drift tube IMS system […]
In the Spring Semester of 2015, Dr. Clowers was recognized within the College of Arts and Sciences at Washington State […]
For those that are interested, here is the spreadsheet used in the ASMS 2018 short course. Thank Dr. Bill Siems […]
We are pleased to announce the unpacking and, more importantly, the successful pump down of the G2. Combined with a […]
Unboxing to commence in t-minus 6 days and counting…
In early 2015 the research group is pleased to bring the next generation ion mobility-ion trap system online. This […]
The Clowers Research Group is now live. Reporting in are the background ions from ionized air measured using a residual […]
Though seemingly outside the realm of chemistry, electronics lie at the heart of many experimental disciplines as all rely ability […]