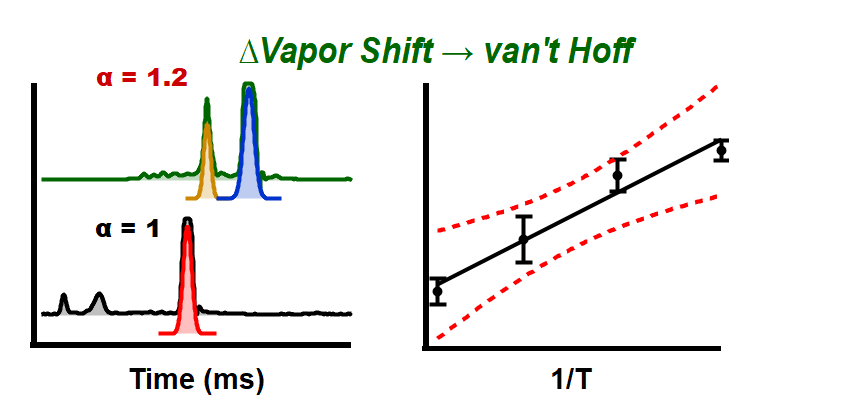
Just accepted in JASMS: Vapor assisted mobility shift measurements were made with atmospheric pressure drift-tube ion mobility–mass spectrometry (IM–MS) to determine the thermodynamic properties of weakly bound ion-molecule clusters formed from protonated phenylalanine and neutral vapor molecules with hydroxyl functional groups. Relative binding energies and gas-phase association energies of amino acid ions clustered with small organic molecules have been established previously using high-pressure mass spectrometry. However, the issue of volatility largely prohibits the use of high-pressure mass spectrometry for the determination of gas-phase associations of amino acid ions clustered with neutral vapor molecules in many instances. In contrast, ion mobility measurements can be made at atmospheric pressure with volatile vapor additives near and above their boiling points, providing access to clustering equilibria not possible using high-vacuum techniques. In this study, we report the gas-phase association energies, enthalpies, and entropies for a protonated phenylalanine ion clustered with three neutral vapor molecules: 2-propanol, 1-butanol, and 2-pentanol based upon measurements at temperatures ranging from 120 to 180 °C. The gas-phase enthalpy and entropy changes ranged between −4 to −7 kcal/mol and −3 to 6 cal/(mol K), respectively. We found enthalpically favored ion-neutral cluster reactions for phenylalanine with entropic barriers for the formation of phenylalanine–1-butanol and phenylalanine–2-pentanol cluster ions, while phenylalanine–2-propanol cluster ion formation is both enthalpically and (weakly) entropically favorable. Under the measurement conditions examined, phenylalanine–vapor modifier cluster ion formation is clearly observed via shifts in the drift time for the three test vapor molecules. In comparison, negligible shifts in mobility are observed for protonated arginine exposed to the same vapor modifiers.
Read the full article here: https://pubs.acs.org/doi/full/10.1021/jasms.0c00020
Comments are closed